Look ahead: Why viability virus testing prevents false alarms
Plant virus viability is the difference between “viral RNA detected” panic and a calm, on-schedule day.
Picture mid-September in Ontario: crews are deleafing and sanitizing for winter production. Two weeks later, a routine drain-water screen flags tomato brown rugose fruit virus (ToBRFV) RNA. Panic? Not this time. A viability assay shows particles are non-infective, the schedule holds, and an eight-week production gap is avoided.
That small data point can be worth hundreds of thousands of dollars in winter revenue – exactly why viability testing belongs on every Canadian grower’s fall checklist.

Viruses: stealthy pathogens that make viability checks essential
Fungi and bacteria can be cultured, plated, and usually controlled with chemistry or biocontrols. Plant viruses are different:
- Obligate parasites: no growth on agar.
- No curative chemistry once systemic infection occurs, infected plants must be culled.
- Long survival on hard surfaces and weeks in water; spread via tools, drain lines, or gloves (even bee hives!) [Viruses][Frontiers in Plant Science].
For this blog, we’ll use ToBRFV as an example. It has now been established in every major Canadian tomato region, overcomes the Tm-22 resistance gene, and infects tomato and pepper [Molecular Plant Pathology][Plant Disease]. That host range makes greenhouses ground zero for the virus.

Learn More about Viability Testing and Pathogen Mini 2.0
More information, like targets and pricing!
Viability virus testing 101: What “positive” should really mean
In the last section, we noted that viruses can linger on hard surfaces for months and move quietly through water and other surfaces, often long after visible symptoms fade. A simple “detected” isn’t enough during fall change-outs; you need to know if particles can still start an infection.
What each test tells you
- Standard qPCR: Think of it as a smoke alarm. It’s very good at sensing viral genetic “smoke,” but it can’t tell if there’s still an active fire. It may pick up harmless leftovers from broken virus particles along with real risks.
- Viability testing: This is a quick add-on step before qPCR that blocks the signal from broken, non-infectious particles. What remains in the result primarily reflects intact particles that could still infect plants.
It’s like checking a light bulb: not just seeing that a bulb is in the box, but screwing it in to confirm it still works. Viability testing helps separate “present” from “still capable of infecting,” and it’s already used in complex samples like wastewater and foods to reduce false alarms and give a practical, same-day read on risk.
When decisions are high-stakes
For major calls like crop removal or restarting production, some labs also run a simple indicator-plant test. A small amount of sap or water is gently rubbed onto sensitive plants; if small local spots appear, the virus is infectious. It takes longer, but it’s a solid confirmation when the consequences are significant.
Here are papers you can read to learn more:
- Nature Scientific Reports: https://www.nature.com/articles/s41598-022-08452-1
- Journal of Applied Microbiology: https://academic.oup.com/jambio/article/124/4/958/6714013
- Systematic and Applied Microbiology: https://linkinghub.elsevier.com/retrieve/pii/S1438463917302468
- Plants: https://www.mdpi.com/2223-7747/11/24/3443

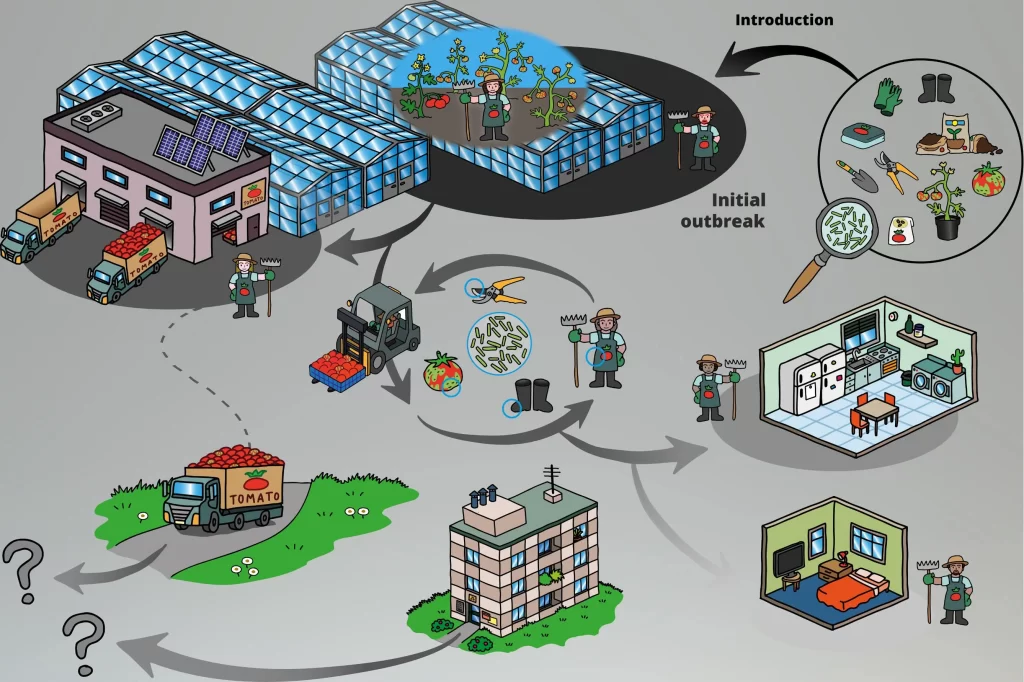
Why Fall Change-Out Is High Risk in Canada
1. Long-Cycle Crop Residue
Canadian tomato and pepper crops often run nine to eleven months, accumulating pruned leaves, root exudates, and biofilm. Fresh research shows ToBRFV lasts atdays to weeks on many surfaces; months possible on non-porous materials under cool, protected conditions [Viruses]. Fall teardown aerosolises that residue just when the house is empty and ventilation is wide open.
2. Water-Mediated Spread
Recirculating nutrient solution is standard in modern Canadian ranges. ToBRFV remains infective in water for up to four weeks at 20 °C and moves readily through hydroponic plumbing [Frontiers in Plant Science]. Unless the loop is disinfected or validated virus-free, the first irrigations of the new crop can seed an outbreak.
3. Cooler Temperatures Slow Symptom Development
Shorter day length and lower winter light suppress metabolism, delaying symptoms. Viruses can smoulder unseen for six to eight weeks, masking a problem until peak winter harvest.
Integrating Viability Virus Testing into Fall SOPs
| Timing | Sample Type | Target Test | Action Threshold |
| Two weeks pre-pull | 50 mL drain water / zone; glove and shear swabs | Pathogen Mini 2.0 (Viability) | Any viable detection → flag zone for intensive sanitation |
| Post-sanitation (Day 0) | Repeat same surfaces + 10 mL fertigation header sample | Pathogen Mini 2.0 (Viability) | “Not detected – viable” required before transplant arrives |
| Seed & tray receipt | 3 g seed subsample or cotyledon wash | Pathogen Mini 2.0 (Viability) | Destroy lot if viable particles found; treat seed if only RNA detected |
| Weeks 2–4 after planting | Composite drain water weekly | Pathogen Mini 2.0 (Viability) | If viable titre >10³ genome copies mL⁻¹ → install inline ozone or PAA shock |
Don’t Ignore the Abiotic Angle
Virus pressure escalates when plants are stressed. SGS Crop Science offers rapid water, nutrient solution, media, and tissue analyses that help dial in EC, pH, and Ca:K ratios. Proper ion balance strengthens cuticle integrity and can reduce mechanical transmission risk. Think of SGS data as the environmental “weather report” that frames every IPM decision.
Practical Tips for the 2025 Fall Cycle
- Map your greenhouse hydraulics – Dead legs and bypass lines are prime virus reservoirs.
- Assign a “clean crew” – Teams that finish teardown must not enter propagation zones without a 24-h gap and fresh clothing.
- Pair viability data with SGS nutrient reports – High chloride or low calcium often coincide with cracked fruit and higher mechanical transmission.
- Archive all Ct and viability results – Trending data pinpoints chronic hotspots and supports ongoing IPM efforts
Where to get help (and how to ask for viability virus testing)
- Healthy Hydroponics: Targeted pathogen testing with an optional viability upgrade (Pathogen Mini 2.0). Current viability targets include most viruses/viroids such as ToBRFV, CGMMV, PepMV, PMMoV, PVY, HLVd, INSV, ToCV/TICV, and others (confirm per target). More targets like Fusarium, Pythium, Ewinia, and Neopestalotiopsis will come very soon. Same sample types as standard testing (water, substrate/media, plant tissue, swabs); typical turnaround is within three business days of receipt in their Growers’ Portal.
- SGS Crop Science: Comprehensive abiotic and chemical diagnostics (water, nutrient solution, media, tissue) that underpin disease management, fertigation optimization, and environmental conditions that influence transmission risk. SGS also offers broader pathogen panels (non-viability).
Combined, they deliver a complete snapshot of greenhouse health.
Take-Home Message
Treat fall teardown like a controlled biosecurity event. Require viability “not detected” before transplant, validate the recirculation loop, and spot-check drain water in weeks 2–4. Let viability virus testing drive actions: shock, purge, or proceed. Paired with SGS water/nutrient data, you remove both the virus and the stress that helps it move, so you enter winter with clean plants and predictable packs.
Frequently Asked Questions
Some quick answers about viability virus testing for greenhouses and indoor agriculture.
What is viability virus testing?
An added step before qPCR that blocks signals from broken, non-infectious particles, so results focus on intact, potentially infectious virus.
How is it different from standard qPCR?
Standard qPCR can’t distinguish harmless RNA fragments from real risk; viability virus testing does, reducing false alarms during change-outs.
When should I use it?
Anytime you doubt the health of your system. Two weeks pre-pull, post-sanitation, on seed/tray receipt, and weekly in weeks 2–4 after planting—especially in recirculating systems.
Which samples work best?
Plant samples. Drain water, fertigation headers, surface swabs (gloves, shears, gutters), seed subsamples, and cotyledon washes.
What if viability is positive?
Escalate sanitation, validate the loop (ozone/PAA), and retest until the target is not detected.

Need further assistance?
Need help finding the answers you need? Let’s have a conversation.
References
Ehlers, J., Nourinejhad Zarghani, S., Liedtke, S., Kroschewski, B., Büttner, C., & Bandte, M. (2023). Analysis of the spatial dispersion of tomato brown rugose fruit virus on surfaces in a commercial tomato production site. Horticulturae, 9(5), 611. https://doi.org/10.3390/horticulturae9050611
Fougere, G. C., Xu, D., Gaiero, J. R., McCreary, C., Marchand, G., Despres, C., Wang, A., Fall, M. L., & Griffiths, J. S. (2025). Genomic diversity of Tomato brown rugose fruit virus in Canadian greenhouse production systems. Viruses, 17(5), 696. https://doi.org/10.3390/v17050696
Lizana, X., López, A., Benito, S., Agustí, G., Ríos, M., Piqué, N., Marqués, A. M., & Codony, F. (2017). Viability qPCR, a new tool for Legionella risk management. International Journal of Hygiene and Environmental Health, 220(8), 1318–1324. https://doi.org/10.1016/j.ijheh.2017.08.007
Mehle, N., Bačnik, K., Bajde, I., Brodarič, J., Fox, A., Gutiérrez-Aguirre, I., Kitek, M., Kutnjak, D., Loh, Y. L., Maksimović Carvalho Ferreira, O., Ravnikar, M., Vogel, E., Vos, C., & Vučurović, A. (2023). Tomato brown rugose fruit virus in aqueous environments—Survival and significance of water-mediated transmission. Frontiers in Plant Science, 14, 1187920. https://doi.org/10.3389/fpls.2023.1187920
Nourinejhad Zarghani, S., Monavari, M., Ehlers, J., Hamacher, J., Büttner, C., & Bandte, M. (2022). Comparison of models for quantification of tomato brown rugose fruit virus based on a bioassay using a local lesion host. Plants, 11(24), 3443. https://doi.org/10.3390/plants11243443
Randazzo, W., Piqueras, J., Rodríguez-Díaz, J., Aznar, R., & Sánchez, G. (2018). Improving efficiency of viability-qPCR for selective detection of infectious HAV in food and water samples. Journal of Applied Microbiology, 124(4), 958–964. https://doi.org/10.1111/jam.13519
Skelton, A., Frew, L., Ward, R., Hodgson, R., Forde, S., McDonough, S., Webster, G., Chisnall, K., Mynett, M., Buxton-Kirk, A., Fowkes, A. R., Weekes, R., & Fox, A. (2023). Tomato brown rugose fruit virus: Survival and disinfection efficacy on common glasshouse surfaces. Viruses, 15(10), 2076. https://doi.org/10.3390/v15102076
Stobnicka-Kupiec, A., Gołofit-Szymczak, M., Cyprowski, M., & Górny, R. L. (2022). Detection and identification of potentially infectious gastrointestinal and respiratory viruses at workplaces of wastewater treatment plants with viability qPCR/RT-qPCR. Scientific Reports, 12, 4517. https://doi.org/10.1038/s41598-022-08452-1
Zhou, J., Gilliard, A., & Ling, K.-S. (2024). Tomato brown rugose fruit virus is transmissible through a greenhouse hydroponic system but may be inactivated by cold plasma ozone treatment. Horticulturae, 10(4), 416. https://doi.org/10.3390/horticulturae10040416
Banner image from https://commons.wikimedia.org/wiki/File:TMV_virus_under_magnification.jpg
Disclaimer
The information presented in this blog is based on collating published peer-reviewed scientific literature and sources that we consider reliable. This is by no means an exhaustive review of pathogens and IPM strategies. This blog provides a brief overview of what is known about pathogens. We encourage growers to do more research on the pathogens concerning their crops and hydroponic systems. We are not plant pathologists; therefore, the information presented should not be used as professional advice for treating pathogens or operating your system.




